How to register an insecticide for Import
By Team Metacorp
Legal & Compliance Experts
Introduction
- Importing an insecticide into India—even when it is already registered or approved elsewhere—is a tightly regulated activity governed by a combination of biosafety, plant quarantine, and environmental protection laws. The regulatory framework ensures that any insecticide, germplasm, transgenic material, or genetically modified organism (GMO) entering the country does not pose risks to human health, crops, biodiversity, or the agricultural ecosystem.
- For importers, research institutions, universities, and agri-input companies, understanding this approval pathway is essential because the import of such materials is not treated merely as a commercial transaction. It is considered a biosecurity-sensitive activity that must comply with the Plant Quarantine (Regulation of Import into India) Order, 2003, and, where applicable, the Environment (Protection) Act, 1986 and biosafety rules related to genetically engineered organisms.
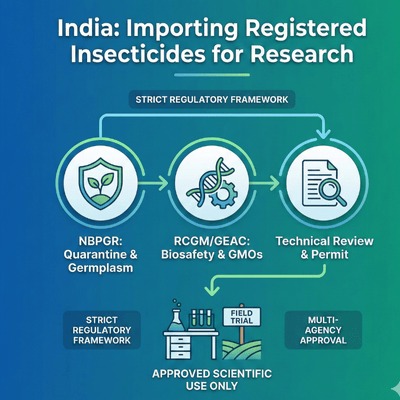
- The process involves multiple authorities, most importantly the Director of the National Bureau of Plant Genetic Resources (NBPGR), who examines the scientific purpose, safety compliance, and quarantine arrangements before granting an import permit. For transgenics or GMOs, additional approvals from Review Committee on Genetic Manipulation (RCGM) and the Genetic Engineering Approval Committee (GEAC) are mandatory.
1. Key Regulatory Authorities
a) National Bureau of Plant Genetic Resources (NBPGR)
NBPGR is the central authority for importing plant germplasm and research materials. It screens applications, verifies research objectives, evaluates quarantine risks, ensures non-commercial use, and coordinates inspection and post-entry monitoring.
b) Review Committee on Genetic Manipulation (RCGM) – DBT
RCGM oversees all GMO-related research. It reviews genetic manipulation data, laboratory containment levels, biosafety protocols, and assesses potential environmental risks before granting research approvals.
c) Genetic Engineering Approval Committee (GEAC)
GEAC, under the Environment (Protection) Act, 1986, evaluates genetically engineered organisms/products, approves their import, export, field trials, or controlled release, and enforces biosafety and environmental safeguards.
d) ICAR, SAUs & International Research Centres
These research institutions act as applicants/collaborators in trials involving insecticides or biological materials. Their internal Institutional Biosafety Committees (IBCs) participate in reviewing, endorsing, and forwarding applications for regulatory approvals.
2. Applicability of the Import Approval
This approval applies to the import of items listed under Schedule-V, Schedule-VI, and Schedule-XII of the Plant Quarantine (Regulation of Import into India) Order, 2003.
It is required when the import is intended for non-commercial scientific use, including:
- Field trials
- Laboratory research
- Greenhouse evaluation
- Breeding programmes
- Development of new formulations
- Comparative efficacy or resistance studies
Note: This approval does not apply to commercial-scale insecticide imports meant for sale or distribution. Such cases require separate processing under GEAC guidelines and registration under the Insecticides Act, 1968.
3. Who Can Apply?
The following categories of applicants are eligible to seek approval for the import of materials covered under Schedule-V, Schedule-VI, and Schedule-XII of the Plant Quarantine (Regulation of Import into India) Order, 2003:
a) Importers
Any individual or company holding valid business registration and authorized to engage in the import of agrochemicals, biological materials, germplasm, transgenics, or related research inputs. The applicant must possess the necessary statutory licences required under applicable Indian laws.
b) Authorised Agents
Legally appointed representatives or agents who have been formally empowered by an importer to act on their behalf. Such agents submit applications, coordinate documentation, and comply with regulatory requirements as per the authorization issued to them.
c) Research Institutions
Eligible research-based entities involved in agricultural, biological, or environmental studies, including:
- ICAR institutes
- State Agricultural Universities (SAUs)
- Accredited laboratories
- Public-sector research and development (R&D) units
These institutions typically import research materials for experimentation, field evaluation, breeding programmes, or laboratory analysis.
d) International Scientific Organisations
Recognized global research bodies engaged in collaborative programmes with Indian agencies, such as:
- ICRISAT (International Crops Research Institute for the Semi-Arid Tropics)
- Other international agricultural research organizations participating in bilateral or multilateral scientific collaborations.
These organisations are permitted to import germplasm, biological agents, or experimental material for joint research initiatives with Indian partners.
4. Pre-Application Documentation
A. Technical Information
- AI name
- Chemical composition
- Formulation type
- Purity specifications
- Manufacturer & country of origin
B. Safety Documents
- MSDS
- CoA
- Basic storage/handling precautions
C. Research Proposal
- Objective of study
- Type of experiment (lab/greenhouse/field)
- Methodology outline
- Duration of study
- Disposal plan
D. Containment & Quarantine Compliance
- Facility details
- Safety measures
- Trained personnel list
E. Legal & Institutional Approvals
- IBSC approval (only if GM material)
- Authorization letter for agent
- Undertaking of non-commercial use
F. Shipping & Logistics Information
- Mode of transport
- Packaging details
- Port of entry
5. Step-by-Step Application Procedure
Step 1: Prepare the Application Dossier
Compile all mandatory technical, safety, research, and quarantine documents. Only complete dossiers are accepted.
Step 2: Submit Application to the Director, NBPGR
Submit as per the current NBPGR format, including:
- Cover letter
- Application form
- Dossier
- Required approvals
- Authorization letter
Step 3: NBPGR Preliminary Scrutiny
NBPGR checks completeness, purpose of import, schedule category, and quarantine readiness, Clarifications requested.
Step 4: Technical Review
Authorities ask for additional information or verify research facility preparedness.
Step 5: Permit Issuance
NBPGR issues the import permit with conditions on quantity, validity, research site, and mandatory quarantine/safety requirements.
Step 6: Port-of-Entry Verification
Plant Quarantine officials check documents and inspect the consignment on arrival.
Step 7: Post-Import Compliance
Importer must use the material strictly for approved research, maintain records, follow disposal norms, and submit reports.
6. Typical Conditions Mentioned in the Permit includes:
- Usage restricted only to the stated trial
- No sale, distribution or commercial use
- Mandatory handling by trained personnel
- Secure laboratory or greenhouse setup
- Submission of research findings
- Disposal as per biosafety guidelines
- Quarantine inspection before use
Conclusion
Importing an insecticide into India—whether or not it is registered abroad—requires strict compliance with national biosafety and plant quarantine regulations. The process is intentionally rigorous to protect India’s agriculture, prevent pest or pathogen entry, and ensure that research materials are used responsibly.
NBPGR oversees the approval pathway, allowing imports only when the scientific purpose is justified and supported by complete technical documentation, appropriate biosafety clearances (such as RCGM/GEAC for GM materials), and adequate quarantine arrangements. Each stage of review—from dossier examination to facility verification and port-of-entry inspection—ensures that all phytosanitary and environmental risks are carefully evaluated.
For more details, please visit Metacorp

